Perhaps you should not seek out antibiotics for acne as soon as you start noticing pimples.
Antibiotics are generally only appropriate for the most severe of acne, which is acne often accompanied by deep scarring, high levels of pain, and acne that doesn't respond to topical treatment options. Otherwise, the drawbacks that come with antibiotics (microbial drug resistance, primarily) mean that they can cause more harm than good.
In most cases, developing and sticking to a thorough and quality skincare routine can significantly improve your acne. Adding antibiotics into the mix, therefore, is often unnecessary, and even risky. Always take the time to exhaust your other options before considering medications, like antibiotics or birth control, for your acne, as you're likely to find that effective topical treatments can do the job without exposing you to any of the unwanted side effects.
Also read: How to choose the best acne treatment
Do Antibiotics Help Acne?
Antibiotics tend to help more than they harm only when your skin doesn't respond to other, less vigorous treatment options.

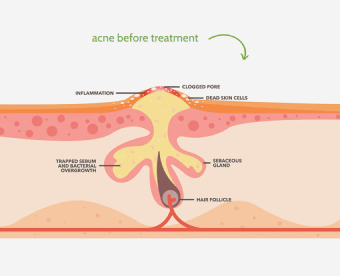
When prescribed for acne, antibiotics primarily work to reduce excess bacteria that cause acne (P. acnes bacteria) and establish a better balance on the skin.
However, taking a step back, antibiotics can pose some serious risks to those who use them. That's why it's important to recognize that you likely don't need to try antibiotic pills. There's no need to set yourself up for long-term consequences unless you've tried all of your other options.
All About Antibiotic Resistance
Antibiotic resistance is perhaps the most pressing risk to consider. When we use antibiotics, we kill not just "bad" bacteria on the skin, but also good bacteria that help the skin function normally.
Antibiotics can not distinguish between "target" bacteria and others, leading to what is essentially a mass wipe-out of all bacteria they encounter.

The bacteria that are left behind, in some cases, are those that are resistant to the drug. In other words, the fittest survive the attack, but these resistant bacteria can reproduce and continue to cause problems.
Eventually, more and more of the bacteria on the skin that causes acne will be resistant to these antibiotics, at which point your antibiotics will no longer be effective.
Even more concerning is the fact that with enough time to develop several resistance mechanisms (ways to resist antibiotics and what they do), bacteria can become immune to not just one specific antibiotic, but all antibiotics.
This has serious implications for treating bacterial infections elsewhere in the body, many of which can lead to severe complications without antibiotics.
Can Antibiotics Make Acne Worse?
Antibiotics have side effects that, in most cases, can exacerbate acne or make it harder to manage. As mentioned above, if the bacteria on your skin becomes resistant to antibiotics, then it could create an infection that no antibiotic can solve.
Antibiotic resistance can cause problems not just on your skin, but in the rest of your body as well.

Side Effects of Antibiotics for Acne
Topical antibiotics may occasionally cause temporary side effects like:
-
Dry skin
-
Irritation
-
Mild flaking of the skin
-
Allergic reactions, which may lead to contact dermatitis
Oral antibiotics for acne, on the other hand, can produce side effects such as:
-
Vomiting
-
Diarrhea
-
Yeast infections
-
Allergic reactions, which may lead to anaphylaxis
What to Use Instead of Antibiotics for Acne
So, if you can't turn to antibiotics for acne, what should you do instead to manage skincare concerns and combat bacteria? Ingredients like benzoyl peroxide, salicylic acid, and more may be the solutions you need.
Create a Thoughtful Topical Skincare Routine
Generally, all acne-prone skin can be improved when treated gently and consistently. Specialized produts like our spot treatments and serums can help you address specific concerns or target certain areas of your skin.
Our products use the power of benzoyl peroxide to kill excess bacteria without disrupting the balance on your skin or contributing to antibiotic resistance. Benzoyl peroxide also kills bacteria that are deep within the pores to prevent future breakouts.

To take advantage of what benzoyl peroxide combined with other useful ingredients, like salicylic acid, has to offer, we recommend the Exposed Basic Kit. Our products include all of the ingredients listed above and more, and we guarantee that following our simple three-step process will significantly improve your skin condition.

When to See a Dermatologist
Though antibiotics aren't generally an advisable acne treatment, there are some cases where their use may be necessary, so much so that the risks they pose are less pressing than the risks of not treating the acne.
However, these cases are very rare.
If your acne is not cystic and isn't causing deep scarring, you probably don't have acne that's so severe that it warrants professional intervention.
On the other hand, if you experience severe, cystic acne or acne inversa, it may be necessary to consult a dermatologist to determine the best method of treatment (regardless of whether it involves antibiotics or not).
Signs you may be dealing with severe cystic acne include:
-
Deep, painful, and large blemishes that may or may not appear to be filled with pus
-
Heavy inflammation, which may lead to very red, swollen skin
-
Significant scarring
-
Itching or pain
-
A lack of improvement or change over time, even after exhausting all of your other skincare options
Likewise, acne inversa, a chronic inflammatory condition, may cause severe blemishes that don't respond to traditional acne treatment because the root of the issue is far more complex than it is for acne vulgaris.
If you do suspect your acne goes above and beyond what's "normal," and have tried everything you can to manage it yourself, it may be necessary to visit a dermatologist and, in some cases, utilize antibiotics.
Still, it's important to be sure that your doctor's treatment plan adheres to American Academy of Dermatology guidelines — patients should take antibiotics for the shortest time possible.
Antibiotics For Acne FAQs
Q: What are the most commonly prescribed antibiotics for acne treatment?
When it comes to treating acne, tetracyclines like doxycycline and minocycline are often the go-to antibiotics, along with erythromycin and clindamycin.

Q: What are the most common side effects of oral antibiotics for acne?
Oral antibiotics for acne can cause a few side effects, including an upset stomach, diarrhea, and changes in appetite. While these side effects are generally mild, in rare cases, some people may experience an allergic reaction or even a serious condition like a C. diff infection.
Q: Are oral antibiotics effective against severe acne?
Yes, oral antibiotics can be effective against severe acne, but they're usually used in combination with other acne treatments. They help to reduce inflammation and kill the bacteria that cause breakouts, ultimately leading to clearer skin.
Q: When should you not treat acne with oral antibiotics?
Oral antibiotics are not suitable for all acne cases. Mild acne doesn't usually require antibiotics, as this can lead to antibiotic resistance. If you have a history of allergic reactions to antibiotics or certain medical conditions, your doctor may advise against using oral antibiotics for your acne.
Q: Are oral antibiotics for acne prescribed together with topical retinoids?
Yes, a combination of oral antibiotics and topical retinoids can be effective for treating acne. Topical retinoids work by unclogging pores and reducing inflammation, while oral antibiotics kill the bacteria that cause acne.

Q: How do I know if the oral antibiotics for acne are working?
It usually takes several weeks to see improvements in your acne with oral antibiotics. If you're on antibiotics, your doctor will monitor your progress and may adjust your treatment plan accordingly.
Q: Are oral antibiotics for acne safe for pregnant women?
In general, oral antibiotics are not recommended for pregnant women as certain antibiotics can harm the developing fetus. If you're pregnant or planning to become pregnant, speak with your doctor about alternative acne treatment options.
Q: Are oral antibiotics safe for teens?
Oral antibiotics are generally safe for teens, but they should only be used as directed by a healthcare professional to prevent antibiotic resistance.
Q: Can oral antibiotic treatment worsen acne?
In some cases, oral antibiotic treatment can cause a flare-up of acne by disrupting the natural balance of bacteria on the skin. If you experience an acne flare-up while on antibiotics, speak with your doctor about adjusting your treatment plan.
Q: Is it safe to treat acne lesions with topical antibiotics?
Topical antibiotics can be effective in treating acne lesions, but they should be used with caution. While topical antibiotics are generally considered safe, they can cause side effects like skin irritation, dryness, and redness.
Additionally, overuse of topical antibiotics can contribute to antibiotic resistance, making it harder to treat bacterial infections in the future.